|
9/17/2023 0 Comments Carbon dioxide chemical formula 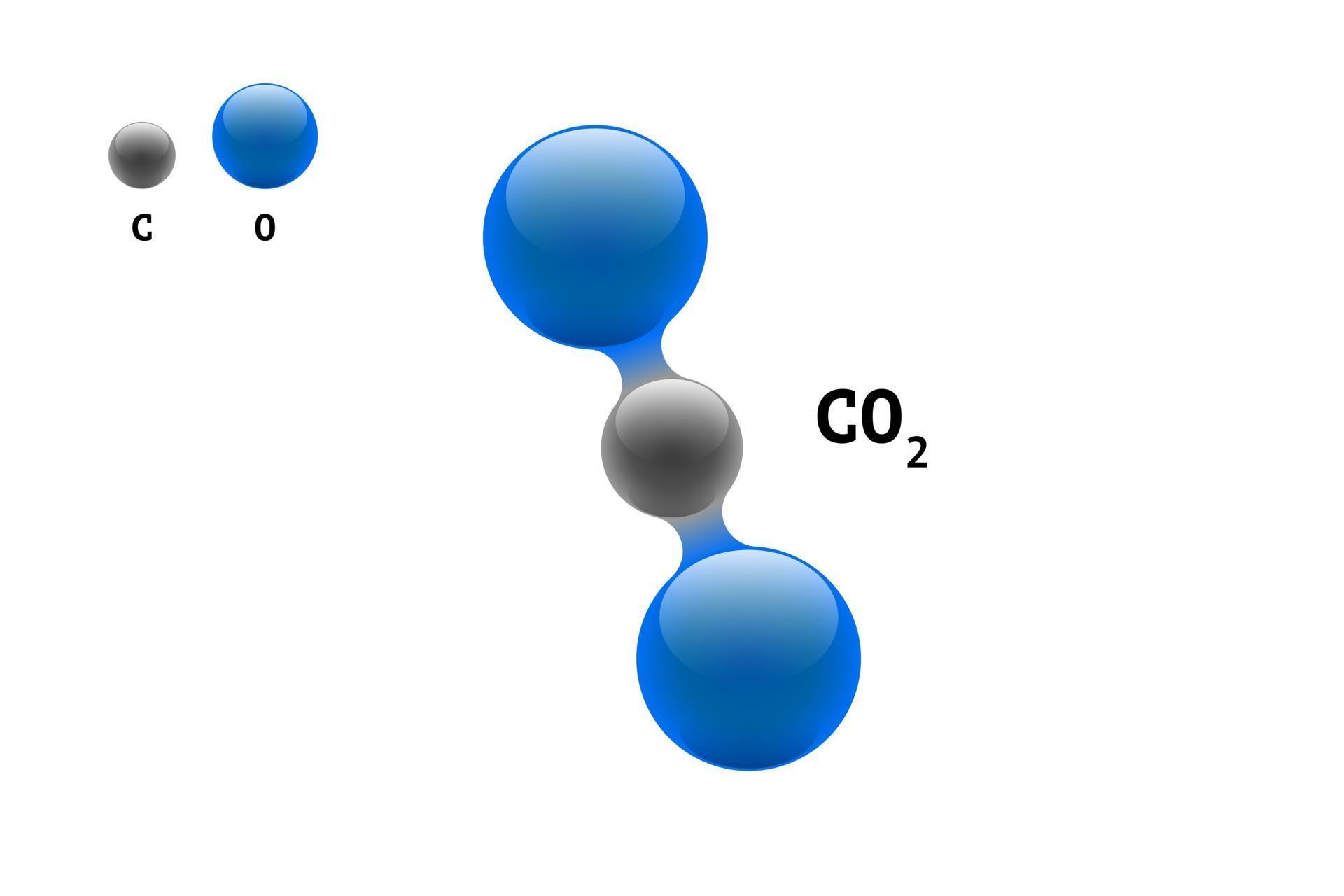
The numbers of ions in a formula must give an equal number of positive and negative charges. The formula of an ionic compound can be predicted by using the formulae of its ions. HCO 3 + H 2 O H 3 O + + CO 32 However, the acid-base behaviour of carbonic acid depends on the different rates of some of the reactions involved, as well as their dependence on the pH of the system. Names and formulae of some common ions: Name of ion The number and sign of its electrical charges are shown in superscript text. IonsĪn ion is a charged particle formed when an atom, or a group of atoms, loses or gains electrons. ChEBI Name carbon dioxide: ChEBI ID CHEBI:16526: Definition A one-carbon compound with formula CO 2 in which the carbon is attached to each oxygen atom by a double bond. They usually (but not always) contain at least one metal element and one non-metal element. Ionic compounds are made up of atoms joined together by ionic bonds. Balanced Chemical Equation for Baking Soda and Vinegar Reaction. If students have not yet met the compositions of. Long-term measurements of CO2 in the atmosphere are conducted at Manua Loa, Hawaii and several international. 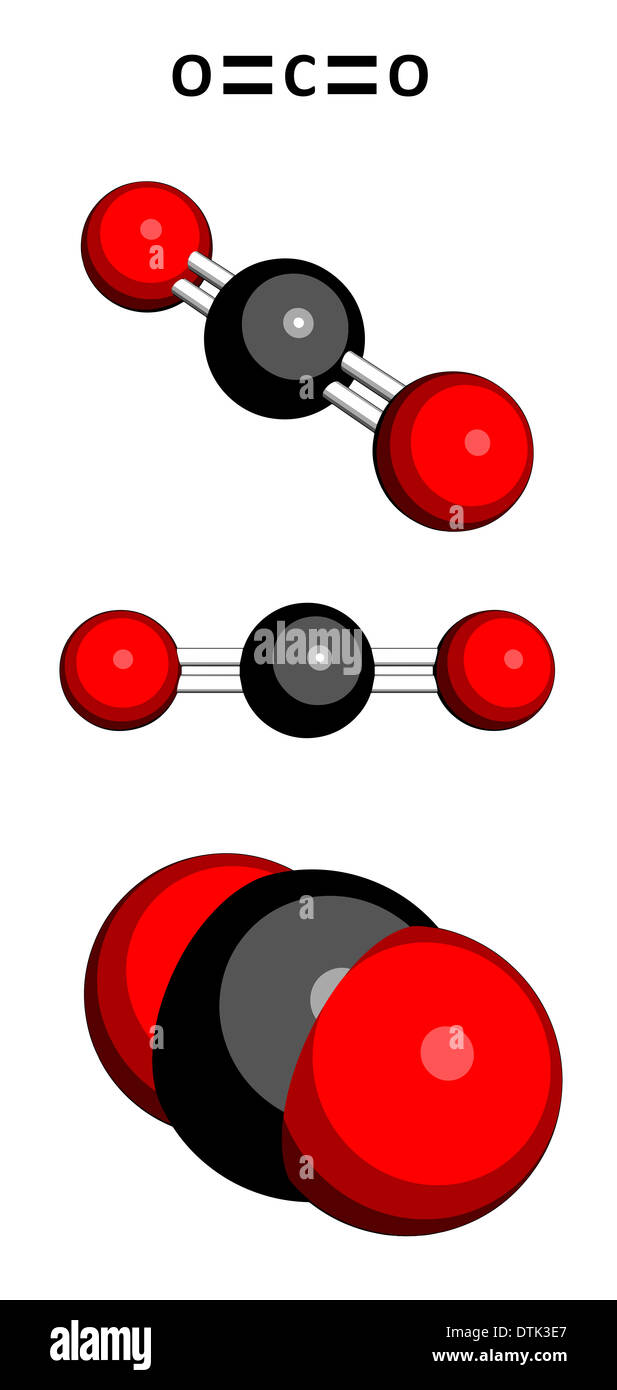
Ionic compoundsĪ bond formed between two atoms where an electron is transferred from one atom to the other is called an ionic bond. The equation for the reaction between carbon dioxide and water may be introduced for appropriate students. The chemical formula for carbon dioxide is CO2. The most common source of Carbon monoxide is thermal combustion. Carbon dioxide is an important greenhouse gas that helps to trap heat in. Carbon monoxide comprises each atom of carbon and oxygen its chemical formula is given as CO. So, the formula NH 3 shows that ammonia contains one nitrogen and three hydrogen atoms. Sulfur Dioxide SO2 or O2S CID 1119 - structure, chemical names, physical and chemical properties, classification, patents, literature, biological activities. A molecule of carbon dioxide (CO2) is made up of one carbon atom and two oxygen atoms. Write a balanced chemical equation for the complete combustion of methane (CH 4 ) with oxygen (O 2 ).The subscript numbers in formulae show how many atoms of that element appear in the molecule. Write the electron-dot formula of carbon dioxide, CO2. Reactants: 2 × C, (2 × O) + (2 × O) = 4 × O. What is the chemical formula for one molecule that consists of 12 carbon atoms, 22 hydrogen atoms, and 11 oxygen atoms Write the formula for the anhydride of H2CO3. poisonous, deadly gas) and carbon dioxide (CO, a greenhouse gas that we exhale. StepĬarbon monoxide + oxygen → carbon dioxide Carbon dioxide, a carbon oxide compound, has a chemical formula of CO 2 and a chemical formula of 44.0095. How are the chemical formula and name of a molecular compound related. The MSDS for propane gives it a health rating of 1, a flammability rating of 4, and reactivity rating of 0. Propane is a three carbon alkane (acyclic saturated hydrocarbon). Write the balanced chemical equation for this reaction. Chemical formula for propane is CH-CH-CH. Repeat steps 3 and 4 until the chemical equation is balanced.Ĭarbon monoxide reacts with oxygen to produce carbon dioxide. You may need to multiply both sides if the number of an atom on the right is not a multiple of the atoms on the left. In this video we'll write the correct formula for Carbon Dioxide.To write the formula for Carbon Dioxide we’ll use the Periodic Table and follow some simple. If necessary, change the number of one of the molecules to make the atoms of at least one element balance. Write a simple equation by writing the name of each substance with its symbol or formula.Ĭheck whether the equation is balanced by counting the atoms of each element. To make the chemical equation correct it must be balanced: Step In a chemical equation, if the names of substances are replaced by their symbols and formulae, most reactions are not correct - there is a different number of atoms in the reactants than in the products. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
